Hãy nhập câu hỏi của bạn vào đây, nếu là tài khoản VIP, bạn sẽ được ưu tiên trả lời.

\(n_{Fe}=a\left(mol\right),n_{Mg}=b\left(mol\right)\)
\(m_{hh}=56a+24b=10.16\left(g\right)\)
\(n_{H_2}=\dfrac{5.6}{22.4}=0.25\left(mol\right)\)
\(Fe+2HCl\rightarrow FeCl_2+H_2\)
\(Mg+2HCl\rightarrow MgCl_2+H_2\)
\(n_{H_2}=a+b=0.25\left(mol\right)\left(2\right)\)
\(\left(1\right),\left(2\right):a=0.13,b=0.12\)
\(m_{Fe}=0.13\cdot56=7.28\left(g\right)\)
\(m_{Mg}=0.12\cdot24=2.88\left(g\right)\)
\(n_{HCl}=2\cdot n_{H_2}=2\cdot0.25=0.5\left(mol\right)\)
\(C_{M_{HCl}}=\dfrac{0.5}{0.5}=1\left(M\right)\)

a)
Gọi : \(\left\{{}\begin{matrix}n_{Al}=a\left(mol\right)\\n_{Fe}=b\left(mol\right)\end{matrix}\right.\)⇒ 27a + 56b = 1,66(1)
\(2Al + 6HCl \to 2AlCl_3 + 3H_2\\ Fe +2 HCl \to FeCl_2 + H_2\)
Theo PTHH :
\(n_{H_2} = 1,5a + b = \dfrac{1,12}{22,4} = 0,05(2)\)
Từ (1)(2) suy ra a = 0,02 ; b = 0,02
Vậy :
\(\%m_{Al} = \dfrac{0,02.27}{1,66}.100\% = 32,53\%\\ \%m_{Fe} = 100\% - 32,53\% = 67,47\%\)
a)
\(n_{HCl} = 2n_{H_2} = 0,05.2 = 0,1(mol)\\ \Rightarrow C\%_{HCl} = \dfrac{0,1.36,5}{100}.100\% = 3,65\%\)

a) \(n_{H_2}=\dfrac{5,6}{22,4}=0,25\left(mol\right)\)
mCu = mY = 9,6 (g)
Gọi số mol Al, Mg là a, b
=> 27a + 24b = 14,7 - 9,6 = 5,1 (g)
PTHH: 2Al + 6HCl --> 2AlCl3 + 3H2
a-->3a-------->a------>1,5a
Mg + 2HCl --> MgCl2 + H2
b--->2b------->b----->b
=> 1,5a + b = 0,25
=> a = 0,1; b = 0,1
=> \(\left\{{}\begin{matrix}m_{Al}=0,1.27=2,7\left(g\right)\\m_{Mg}=0,1.24=2,4\left(g\right)\\m_{Cu}=9,6\left(g\right)\end{matrix}\right.\)
b) nHCl(PTHH) = 3a + 2b = 0,5 (mol)
=> nHCl(thực tế) = \(\dfrac{0,5.120}{100}=0,6\left(mol\right)\)
=> \(C_{M\left(HCl\right)}=\dfrac{0,6}{0,2}=3M\)
c) \(\left\{{}\begin{matrix}C_{M\left(AlCl_3\right)}=\dfrac{0,1}{0,2}=0,5M\\C_{M\left(MgCl_2\right)}=\dfrac{0,1}{0,2}=0,5M\\C_{M\left(HCldư\right)}=\dfrac{0,6-0,5}{0,2}=0,5M\end{matrix}\right.\)
d) \(n_{Cu}=\dfrac{9,6}{64}=0,15\left(mol\right)\)
PTHH: \(Cu+Cl_2\underrightarrow{t^o}CuCl_2\)
0,15-->0,15
=> \(V_{Cl_2}=0,15.22,4=3,36\left(l\right)\)

a) Gọi số mol Mg, Fe là a, b (mol)
=> 24a + 56b = 11,84
\(n_{HCl}=\dfrac{146.14\%}{36,5}=0,56\left(mol\right)\)
PTHH: Mg + 2HCl --> MgCl2 + H2
a--->2a--------->a----->a
Fe + 2HCl --> FeCl2 + H2
b-->2b-------->b------>b
=> 2a + 2b = 0,56
=> a = 0,12; b = 0,16
=> \(\left\{{}\begin{matrix}\%Mg=\dfrac{0,12.24}{11,84}.100\%=24,324\%\\\%Fe=\dfrac{0,16.56}{11,84}.100\%=75,676\%\end{matrix}\right.\)
b) \(n_{H_2}=a+b=0,28\left(mol\right)\)
=> \(V_{H_2}=0,28.22,4=6,272\left(l\right)\)
c) mdd sau pư = 11,84 + 146 - 0,28.2 = 157,28 (g)
=> \(\left\{{}\begin{matrix}C\%_{MgCl_2}=\dfrac{0,12.95}{157,28}.100\%=7,25\%\\C\%_{FeCl_2}=\dfrac{0,16.127}{157,28}.100\%=12,92\%\end{matrix}\right.\)

$a)PTHH:2Al+6HCl\to 2AlCl_3+3H_2$
$n_{H_2}=\dfrac{5,04}{22,4}=0,225(mol)$
$\Rightarrow n_{Al}=0,15(mol)$
$\Rightarrow \%m_{Al}=\dfrac{0,15.27}{9,45}.100\%\approx 42,86\%$
$\Rightarrow \%m_{Cu}=100-42,86=57,14\%$
$b)$ Theo PT: $n_{HCl}=2n_{H_2}=0,45(mol)$
$\Rightarrow C_{M_{HCl}}=\dfrac{0,45.110\%}{0,5}=0,99M$
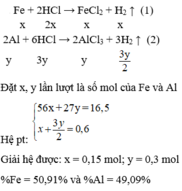



1.
Mg + 2HCl -> MgCl2 + H2 (1)
Fe + 2HCl -> FeCl2 + H2 (2)
nH2=0,08(mol)
Đặt nMg=a
nFe=b
Ta có hệ:
\(\left\{{}\begin{matrix}24a+56b=2,56\\a+b=0,08\end{matrix}\right.\)
=>a=0,06
b=0,02
mMg=0,06.24=1,44(g)
%mMg=\(\dfrac{1,44}{2,56}.100\%=56,25\%\)
%mFe=100-56,25=43,75%
b;
Ta có:
nHCl=2nH2=0,16(mol)
CM dd HCl=\(\dfrac{0,16}{0,1}=1,6M\)